Infants and Children with Tracheostomy and Ventilator Dependence in the Intensive Care Units: Candidacy and Early Intervention with a Bias-Closed, No-Leak Speaking Valve
Laura Brooks, MEd, CCC-SLP, BCS-S
Extensive research on the Passy Muir® Tracheostomy & Ventilator Swallowing and Speaking Valve (PMV®) exists within the adult population to support the benefits of voicing, secretion management, physiologic PEEP, swallowing, olfaction, quality of life, and weaning. However, working with infants and children, who have tracheostomies with or without ventilator support, can be more challenging than with adults due to multiple factors. Developmental factors, in combination with medical concerns, impact treatment considerations, but the research literature in the pediatric population is inadequate to provide sufficient evidence-based practices (Suiter, McCullough & Powell, 2003). Review of recent literature suggests that approximately half of all pediatric patients who receive a tracheostomy are younger than one year of age (Barbato, Bottecchia & Snijders, 2012; Lewis, Carron, Perkins, Sie & Feudtner, 2003). Early tracheostomy may lead to an opportunity for early application of the PMV that may otherwise be missed if the medical team does not have a clear understanding of practice guidelines for PMV application.
Because of the paucity of research in pediatrics, it is challenging to have consensus among physicians and clinicians regarding candidacy for Passy Muir® Valve application with medically complex infants and children. This is particularly difficult for infants in the Neonatal Intensive Care Unit (NICU), patients who are ventilator dependent, and individuals with airway compromise (i.e. stenosis or vocal fold paralysis). As a result, patients who may be a candidate for Valve placement may not receive this intervention due to physician concern for use in what is viewed as a higher risk population.
Therefore, it is critical that the speech-language pathologist has a thorough understanding of the ventilator and the patient’s specific settings, how the PMV changes the mechanics of inspiration and expiration when on the ventilator, and medical co-morbidities that may compromise successful PMV application. The clinicians and facility should have a practice guideline in order to ensure consistent application of the PMV and to provide an understanding of any potential contraindications.
Understanding the Ventilator
The PMV was invented for use in-line with ventilator circuitry (for patients who are ventilator dependent) by a patient who was ventilator dependent. It is a bias-closed, one-way Valve that allows inspiratory support from the ventilator and allows 100% of exhalation to occur out through the patient’s nose and mouth. For best practice, the PMV is typically placed in the ventilator circuit and not directly on the tracheostomy hub. Placement of the PMV on the hub of the tracheostomy tube may create torque. If torque or movement of the tracheostomy tube occurs, there is a higher risk for potential tissue erosion, laceration of the skin, or an exacerbation of granulation tissue growth (Keens, Kun, & Davidson Ward, 2017). Because of the variety of hospital and home ventilators and circuits, clinicians and caregivers must understand the differences between them and the level of support that the patient is receiving from the ventilator.
Some ventilators are designed for use with patients in intensive care units. These ventilators are precise and most frequently used for higher-risk patients, who require more ventilator support. Home ventilators, such as the LTV and Trilogy, are more portable, less expensive, and may be used for patients transitioning from the ICU to the acutecare floor and then to home. Pediatric candidates for home ventilators are children who have relatively stable ventilator settings, with lower FiO2 (<40%) and peak inspiratory pressure (PIP) (<40 cmH2O) (Keens et al., 2017).
When working with patients on mechanical ventilation, an understanding of the ventilator settings and patient parameters is essential for all healthcare professionals. There are two primary types of ventilation: pressure controlled and volume controlled. A physician orders the type of ventilation, depending on the patient’s needs. The following terms are some of the common terms related to the care of a patient on mechanical ventilation with which the healthcare professional should be familiar:
Breath Types:
Volume breath: Ventilator delivers a pre-set volume, regardless of the pressure required to do so. Volume is constant, whereas pressure is
variable (pressure varies depending on lung compliance/resistance).
Pressure breath: Ventilator delivers a pre-set pressure over a pre-set inspiratory time. Pressure is constant, whereas volume is variable (volume varies depending on lung compliance/resistance).
Common Modes of Ventilation:
Pressure Control Ventilation (PC or PC/PS): Ventilator delivers a predetermined number of breaths per minute, with a pre-set pressure over a pre-set inspiratory time. Pressure support may be provided during spontaneous breathing on some ventilators.
Assist Control (A/C): Ventilator delivers a predetermined number of breaths per minute, using either a specified volume or pressure. All triggered breaths are fully supported.
Synchronized Intermittent Mandatory Ventilation with Pressure Support (SIMV/PS): Ventilator delivers a predetermined number of breaths per minute using either a specified volume or pressure. Pressure support is provided during the spontaneous breath.
Pressure Regulated Volume Control (PRVC): Ventilator adjusts the pressure delivered during each breath to ensure target volumes are delivered. Pressure Support with Continuous Positive Airway Pressure (PS w/ CPAP): Continuous positive airway is maintained during exhalation, while each spontaneous breath is supported with a set pressure.
Ventilator Settings (what the physician orders):
Breath types:
- Pressure breaths: Physician orders set pressure.
- Volume breaths: Physician orders set volume.
Positive End-Expiratory Pressure (PEEP): Amount of pressure that remains in the lungs at the end of exhalation.
CPAP: Continuous positive airway pressure.
Pressure Support (PS): Positive pressure provided during a spontaneous breath.
Respiratory Rate (RR): Number of breaths per minute delivered by the ventilator.
Fraction of Inspired Oxygen (FiO2): Percentage of oxygen the ventilator delivers. For reference, room air has FiO2 of 21%.
Tidal Volume (Vt): Volume of gas inhaled with each breath, recorded in cc/ml. Physicians prescribe tidal volume using ideal body weight
and lung pathology.
Other:
Peak Inspiratory Pressure (PIP): Highest level of pressure applied to the lungs during inhalation.
End-Tidal Carbon Dioxide (EtCO2): Capnograph measures exhaled CO2. This value can either be found on the ventilator or on a separate machine. EtCO2 readings may indicate the quality of ventilation or cardiac output and is the gold standard to confirm endotracheal tube placement.
Partial Pressure Carbon Dioxide (PaCO2): Measured from an arterial blood sample. Normal values range from 35-45 mmHg.
Inspiratory Time/I-Time: Duration of inspiration in seconds.
Indications for the Tracheostomy
When working with this patient population, it is important to understand the indications for a tracheostomy. The disease process and reason for tracheostomy may impact the timing of intervention as it relates to PMV use. With infants and children, several causes may lead to a tracheostomy. Three main categories of tracheostomy indications include airway obstruction, lung disease, and neuromuscular/neurological involvement. These categories include, but are not limited to, chronic obstruction within the airway, such as choanal atresia, subglottic stenosis, tracheomalacia, laryngomalacia, and bronchomalacia;vocal cord paralysis, leading to chronic aspiration or poor pulmonary toileting with an inability to clear secretions; severe CNS (Central Nervous System) impairment, such as seen with Arnold-Chiari malformation, Werdnig Hoffmann disease, and Congenital Hypoventilation Syndrome; craniofacial anomalies, such as seen with Pierre Robin sequence and Treacher Collins, Beckwith-Wiedemann, and CHARGE syndromes; and chronic lung disease, including bronchopulmonary dysplasia (Shaker & Mutnik, 2012). Timing of interventions and establishing access to the upper airway for communication, speech-language development, cough, and other pulmonary functions is crucial. Early intervention and use of a PMV provides benefits which may assist in the recovery process.
If the patient has neurologic indications for a tracheostomy, but the lungs are healthy and the muscles are weak, these patients generally do not require frequent changes in ventilator settings (Keens et al., 2017). For patients with upper airway anomalies requiring a tracheostomy, the ability of the patient to adequately exhale around the tracheostomy tube is of concern and would need to be considered during the evaluation. This diagnosis may even require a Direct Laryngoscopy and Bronchoscopy (DLB) to be performed by the otolaryngologist. This assessment would address the severity of the obstruction. Because of the wide variety of causes for a tracheostomy, the history provides crucial information which may impact the assessment process.
Understanding the Impact of a Cuff and Its Proper Management
Generally, uncuffed tracheostomy tubes are the preferred tracheostomy tube type for children. However, patients with severe restrictive lung disease or neuromuscular disease require a high pressure be delivered, and it is done more effectively with the cuff inflated (Hess & Altobelli, 2014). Previously, only uncuffed tracheostomy tubes were available for pediatrics, but in the past decade, cuffed tracheostomy tubes have become more popular (Watters, 2017). The choice of cuffed versus uncuffed tracheostomy tubes is usually institution or patient dependent. The uncuffed tracheostomy tube has benefits not observed in cuffed tracheostomy tubes, such as reducing the incidence of acquired tracheal wall injury (Hess & Altobelli, 2014) and improving phonation (DeMauro et al., 2014; Cowell, Schlossler, & Joy, 2000).
The patient with an uncuffed tracheostomy tube also may have less difficulty with the application of the PMV as there is less change in the exhalation physiology. Typically, a patient inhales and exhales through the tracheostomy tube, which is either cuffed or cuffless. Cuffed trach tubes must be completely deflated prior to PMV application, and the deflated cuff material may still cause some resistance when exhaling (Beard & Monaco, 1993). A tight to the shaft (TTS) tracheostomy tube or uncuffed tracheostomy tube may allow for more space in the tracheal lumen for exhalation out through the mouth and nose. When the PMV is placed, a child still inhales through the Valve and tracheostomy tube, but the Valve closes at the end of inspiration and redirects airflow out through the upper airway, mouth, and nose. For children, the most common reasons for PMV success involve both physiologic and behavioral factors (Lieu, Muntz, Prater, & Stahl, 1999). As such, uncuffed tracheostomy tubes can help prepare the patient physiologically and behaviorally for the change in exhalation. Additionally, an uncuffed tracheostomy tube has the potential to allow the patient to sense the secretions in their pharynx, resulting in a swallow or cough in response. One study with critically ill patients with a tracheostomy, who were randomized to groups, found that deflating the tracheostomy tube cuff shortened weaning time, reduced respiratory infections, and improved swallowing (Hernandez et al., 2013).

Another Consideration: Ventilator Circuits
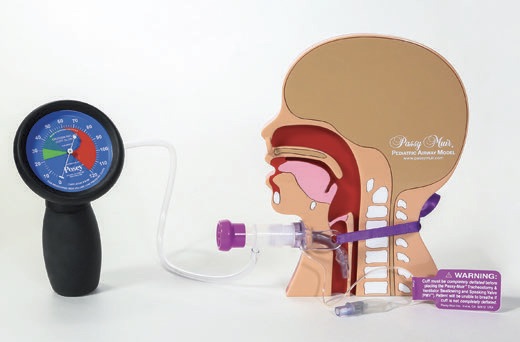
When working with a patient who is ventilator dependent, the speech-language pathologist (SLP) and the respiratory therapist (RT) must be familiar with the different ventilator circuits that may be used. The type of circuitry will dictate the type of adapters that may be needed for successful placement of the PMV in-line with the ventilator circuit. The types of adapters are usually either a 15/22 mm step-down adapter or a 22mm silicone adapter (see Image 1).
It is important to understand the different circuits and know whether the patient is on a single limb circuit or double limb circuit. In addition, the team should be aware if the circuit is a passive circuit or an active
circuit. An example of a ventilator that has both an active and passive circuit that is used often in
pediatrics is the Trilogy. Both circuits are single-limb circuits. The passive circuit has the whisper swivel valve, and the active circuit has a mushroom valve for exhalation. With the passive circuit, the PMV is used with patients who require pressure ventilation. With an active circuit, the PMV is used with patients who are volume ventilated.
The SLP and the RT work together as a team and rely heavily on the expertise and support of the other team members when determining patient candidacy, problem solving ventilator application, and evaluating and treating the patient for Valve use. For successful application and early intervention in critical care, all team members should have extensive understanding of PMV use; otherwise, there may be roadblocks to early application of the Valve on a patient who is ventilator dependent. While the SLP should be educated on ventilator settings, modes, and circuits to help advocate for application of the PMV, the SLP relies on the expertise of the RT for ventilator adjustments and patient safety. The RT relies on the SLP to provide assessment of voice, swallowing, speech and language skills, and cognition.
Application of the PMV: How to Maximize Safety and Success
Understanding the value of the PMV application for patients and the benefits that may be achieved assist with improved patient use and care. However, many patients are underserved due to a lack of clinician and physician consensus for understanding the range of benefits and for determining candidacy. Members of the medical team may ask such questions as: is this patient too young? Too small? Too sick? On too much PEEP? Can the patient tolerate the PMV with any degree of airway obstruction or narrowing?
The benefits of using a bias-closed, one-way valve have been reported in the literature and include access for the infant to be able to communicate via cries and other sounds; have improved taste and smell; generate subglottic pressure for cough, cry, and swallowing; reduce the potential for further vocal cord dysfunction; restore laryngeal/pharyngeal sensation; and improve secretion management (O’Connor, Morris, & Paratz, 2019; Hull, Dumas, Crowley, & Kharasch, 2005; Torres & Sirbegovic, 2004). Abraham (2009) investigated the use of a PMV in children and reported that children wearing a Passy Muir Valve during waking hours normalized secretion management within two weeks due to improved sensation of secretions. Benefits also were reported for reduced time to decannulation and restored physiologic PEEP, which led to diminished WOB (work of breathing) (Hull et al, 2005; Torres & Sirbegovic, 2004; Sutt et al., 2016).
Review of the current literature supports safety of PMV application with certain patients, depending on the medical comorbidities. Passy Muir Valves have been used with both pediatric and adult populations, with the PMV being used with infants as young as one day old and within the NICU (Torres & Sirbegovic, 2004). Some specialists may have concerns that an infant’s airway is too small and will not have enough room around the tracheostomy tube (Torres & Sirbegovic, 2004). However, the concerns related to upper airway patency may be assessed in two different ways: visual observation by the otolaryngologist via DLB and testing with manometry. If it is determined initially that the patient’s upper airway is not patent via endoscopy or manometry, then the infant should be followed and retested, as appropriate, during their admission. Retesting is warranted because an infant or young child may have significant improvement in airway patency secondary to changes in age, weight, or growth which may affect the size of the trachea.
Once it is established that the patient is a good candidate and has a patent upper airway, additional criteria are considered. For Valve placement, the following criteria may be considered for patients who are ventilator dependent:
- a. The patient must tolerate cuff deflation. Set the patient up for success by slowly deflating the cuff. Some patients may even require deflation to take place over several minutes to adjust to the change in airflow (Hess & Altobelli, 2014).
- b. PMV, in the pediatric population, should be trialed following the patient’s first trach change. The first trach change is often done by the surgeon as the immature stoma poses some risk for damage (Hess & Altobelli, 2014).
- c. The patient must be hemodynamically stable.
- d. Contraindications for PMV application:
- i. Significant upper airway obstruction (e.g. grade 4 subglottic stenosis).
- ii. Thick secretions.
- iii. Foam-filled cuff, as these cuffs cannot be safely deflated (Hofmann, Bolton, & Ferry, 2008).
- iv. With the Trilogy ventilators: For the passive circuit, use the PMV with patients who require pressure ventilation. With an active circuit, use the PMV with patients who are volume ventilated.
- e. FiO2 < 50%
- f. PEEP < 10 cmH2O
- g. PIP/PAP= < 40 cmH2O
* some variation exists between facilities (e.g. some
use PEEP of 12 or less).
It is recommended that the medical team continue to apply heated humidification. However, a heat-moisture exchanger (HME) should not be used with the PMV, as no exhaled gas passes to the HME through the tracheostomy tube when the Valve is in place (Hess & Altobelli, 2014).
When using the Passy Muir Valve during mechanical ventilation, respiratory therapists may make some adjustments, under physician direction, to improve patient comfort and safety. Some common and simple adjustments may include:
Reduction or elimination of PEEP: The establishment of a closed respiratory system and exhalation through the oronasopharynx restores physiologic PEEP. This enables the clinician to reduce or eliminate set mechanical PEEP (Sutt et al., 2016). This adjustment may alsoeliminate any unnecessary continuous airflow within the circuit. Continuous flow in the circuit may make it difficult for the patient to close the vocal cords and may stimulate continuous coughing and auto-triggering of the ventilator.
Volume compensation: For patients with inspiratory volume loss, after cuff deflation, additional Tidal Volume (Vt) may be provided until baseline Peak Inspiratory Pressure (PIP) is reached. When considering use of a PMV with mechanical ventilation, factors such as inspiratory support may be managed by ensuring the patient achieves baseline Peak Inspiratory Pressures.
Alarm adjustments: All alarms on the ventilator must be re-evaluated for appropriate adjustments before, during, and after use of the Valve. Proper alarm management is essential for patient safety and best standard of care.
Options for alarm management are dependent upon facility policy. Patient safety is the priority and proper management of the ventilator is key. With clear understanding of the ventilator and the changes that the PMV applies to the respiratory system, the
members of the care team may advocate for adjustments for best practice and improved likelihood of patient satisfaction and comfort (ordered by the physician). It is recommended that a procedure be in place to identify when settings were changed. Proper documentation allows for the ventilator to be returned to the baseline settings when the PMV is removed.
Manometry: Measuring Transtracheal Pressure and Ensuring Airway Patency
To address the issue of the airway and atypical airflow, the step of assessing airway patency with manometry may provide information to the medical team regarding the patient’s ability to exhale adequately around the trachea. If there is obstruction and the patient cannot adequately exhale, pressure can incrementally increase with each breath, known as breath stacking or air trapping (Hess, 2005; Hofmann et al., 2008). Additionally, a higher end-expiratory pressure reading with manometry may indicate patient discomfort, even if the patient is not breath stacking.
For medically complex infants in ICUs, initiating Transtracheal Pressure (TTP) testing as part of every PMV assessment is a helpful tool for objective feedback to physicians and the team regarding safety and readiness for Valve application. Transtracheal pressure testing equipment includes a manometer to be applied within the ventilator circuit with O2 tubing and an adapter. Adapters, such as the 15/22mm step-down adapter or a 22mm silicone adapter (see Image 1), may be added into the circuit as well as aiding proper fit of the Valve. The assessment team, typically respiratory therapy and speech-language pathology, determines how to place the Valve into the circuit, with and without the manometer.
A TTP value is the number at the end of the exhalation or end-expiratory pressure with resting breaths only. This reading provides the patient’s physiologic PEEP (positive end-expiratory pressure). When placing the PMV, a closed system is reestablished which restores a more normal physiologic PEEP, as compared to the PEEP provided by the ventilator. An adequate TTP reading provides feedback to the team that the airway is patent, and the patient may adequately exhale around the tracheostomy tube. The pediatric population presents a special challenge during evaluation because any movement or vocalization will increase the pressure and compromise the ability to read resting breaths. If an infant is crying, moving, vocalizing, or pushing, the pressures will be increased, and it will not be an accurate reading. Challenges with pediatrics occur not only because of the smaller anatomy but due to the difficulties with following specific directions, such as “just breathe” or “don’t move.”
One option to address these issues is to obtain TTP readings while the patient is sleeping in order to test true resting breaths, as the measurement can be taken in as little as 20 seconds. However, the team should consider that despite current literature supporting application of the PMV during sleep (Barraza et al., 2014), use of the Valve during sleep is an off-label use. Alternatives to placing the Valve for TTP measurement during sleep is to catch the child in either a drowsy state or to distract with toys or videos.
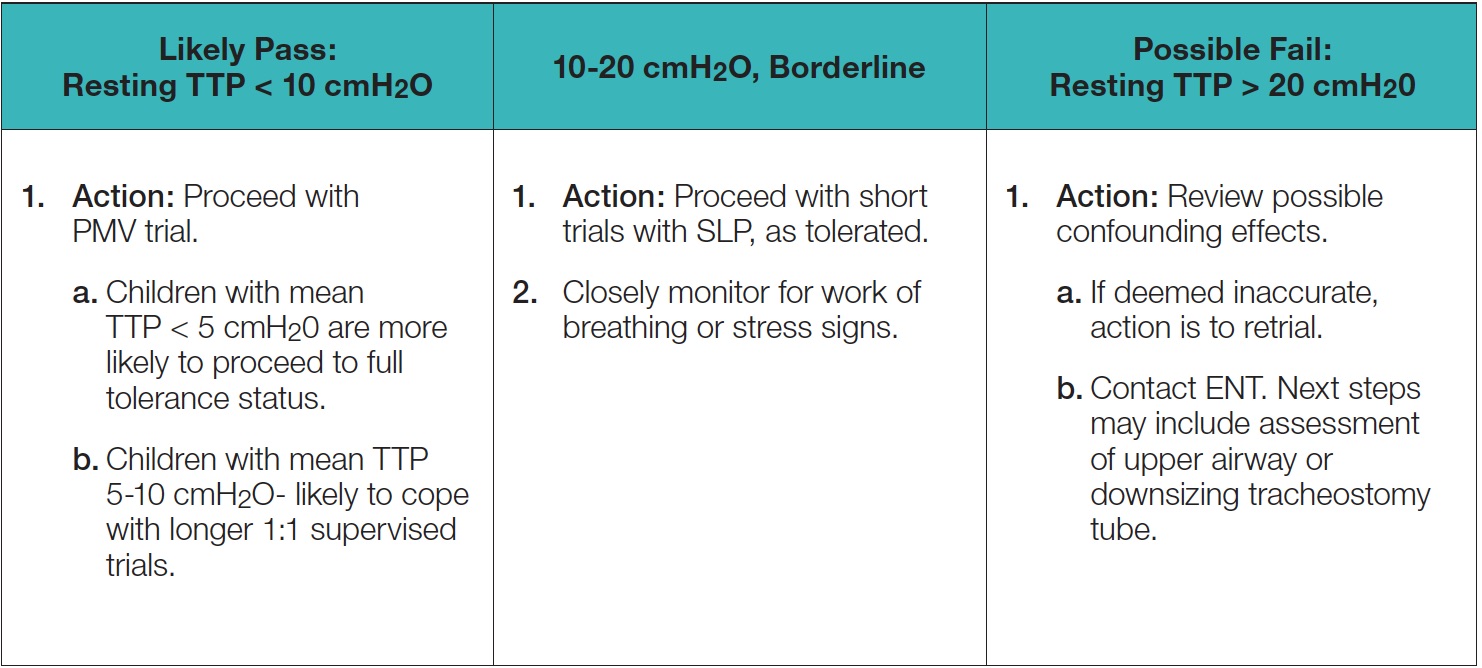
Another consideration is the current discrepancy as to what value is deemed acceptable, meaning what TTP reading or number demonstrates that the airway is patent, and the patient may comfortably and adequately exhale around the tracheostomy tube.An early study suggested that a tracheal pressure greater than 5 cmH2O during passive exhalation may indicate excessive expiratory resistance (Hess, 2005). However, most studies have reported that pressures in the range of 2-6 cmH2O indicate a patent airway and that assessment for use of the Valve may occur(Barraza et al., 2014; Buswell, Powell, & Powell,2016). Additionally, recent research has indicated that children with end-expiratory pressure up to 10cmH2O may tolerate the Valve (Utrarachkij, Pongsasnongkul,Preutthipan, & Chantarojanasri, 2005).In an earlier study, Trotter (1995) found accurate predictions for success with the PMV occurred when patients’ end-expiratory pressures were 15 cmH20 or lower. Trotter also indicated that SpO2 was not a good predictor for Valve use. The literature provides a range of airway patency measurements at which predicting success for Valve use has occurred. The use of TTP is one method for providing an objective measurement that may assist with evaluating patients and may identify potential airway difficulties or even successes. Due to the range of measurements, further research is warranted.
Obtaining an accurate TTP reading requires a good understanding of respiratory and ventilator basics, such as PIP and PEEP, and the differences between ventilators and circuits. Therefore, it may be helpful initially to test airway patency via manometry to obtain baseline measurements without the PMV. Generally, the manometer, without the PMV in place, will read PIP (inspiration) to PEEP (peak endexpiratory pressure) values, which are similar to what is set with the ventilator. For example, if the patient’s ventilator is set to a PEEP of 8 cmH2O and a PIP of 20 cmH2O, the manometer should fluctuate between 8-20 cmH2O with each breath. This consistency may provide a means of calibrating the TTP and identifying accurate readings. Although, at times, the PIP value on the manometer may be slightly lower than the ventilator PIP, such as when there is an exhalation valve, as seen with the Trilogy.
Once the SLP and RT obtain a patient’s manometry baseline, the PMV is placed in-line with the manometer and adapters. With resting breaths only, the TTP reading is the value at the end of exhalation. While this process may seem simple, in actuality, it is challenging to get accurate readings without proper training. The numbers may be misread, especially if a clinician is not familiar with the ventilator, respiratory function, or manometry readings. Importantly, the clinician should not initially read the high number as the inhalation or PIP and the low number as the exhalation or PEEP. In fact, with initial placement, a high number may be the exhalation, but once the patient settles and resting breaths are measured, the high number may be the PIP. It is helpful to watch the baby or child and the manometer for indicators. The RT also contributes information from the ventilator by monitoring inhalation via the ventilator and providing an indication when the patient is at the end of exhalation. Marking the end of exhalation provides a more accurate reading for expiratory pressure. Watching the infant’s chest rise and fall provides relevant information as well. TTP readings may be impacted by position and state, so pressures may need to be retested if the child is moving, agitated, crying, or engaging in other activities that may interfere with readings. Because of the factors that may impact TTP measurements, the SLP and RT may need multiple sessions to get a proper measurement.
If the pressure is too high, breath stacking occurs, or discomfort is visible during exhalation through the nose and mouth, the following should be considered:
It should be noted that even if the airway is patent, other factors can interfere with use of the PMV. Therefore, the SLP and RT must offer the opportunity to use the Valve safely and consistently (Hull, 2005).
A Facility’s Guideline to Passy Muir Valve Application and Best Practice
With limited research on PMV application in the pediatric, medically complex, ventilator-dependent population, it is recommended that facilities develop best practice guidelines for PMV application. Often these guidelines have input from and are approved by pulmonology, otolaryngology, respiratory therapy, and speech-language pathology, among other specialties. To provide best practice and state-of-the-art care for the medically complex, pediatric patient with tracheostomy or ventilator dependence, it is essential that the clinical professionals be familiar with all aspects of respiratory function, including appropriate interventions and assessments, to enhance access to and use of the PMV.
This sample guideline provides suggested steps for patient selection; proper ventilator and alarm considerations; and assessment and application processes for use of the PMV in the pediatric patient population:
I. PROCEDURE:
- A. Criteria for candidacy:
- a . Placement after first trach change.
- b . Tolerance of cuff deflation.
- c . Being hemodynamically stable.
- d . Physician to review the most recent airway examination and determine if follow up is needed, before Valve placement.
- e . Physician to consider indication for tracheostomy, size of tracheostomy tube, and upper airway obstruction to determine if a patient is a candidate for Valve placement.
- f . Patient’s age and weight.
- g . Contraindications:
- i. Significant upper airway obstruction per ENT or pulmonology.
- ii. Copious, thick secretions.
- iii. Foam-filled cuff.
- iv. Airway stenting.
- B. Ventilator parameter recommendations for candidacy:
- a. FiO2 < 50%
- b. PEEP < 10 cmH2O
- c. PIP/PAP < 40 cmH2O
- C. Application of PMV for patients who are on a ventilator.
- a. Physician to order:
- i. Passy Muir Valve trial (Respiratory order) through SLP consult.
- ii. SLP conducts bedside evaluation, in conjunction with respiratory therapy.
- b. Supplies for in-line placement
- c. Pressure testing supplies (see Image 2).
- d. Position patient upright.
- e. Observe baseline vitals.
- f. Oral care and suctioning, as needed.
- g. RT to:
- i. Deep suction tracheostomy, if needed.
- ii. Observe PIP and exhaled Vt.
- iii. Deflate cuff slowly.
- i v. Suction trach and mouth again, as needed.
- v. Look for loss of exhaled Vt.
- vi. Observe changes in vitals, color, work of breathing, and signs of stress.
- h. Proceed, if tolerating the above steps.
- i. Apply PMV and transtracheal pressure manometry in-line with the ventilator circuitry (not directly to the tracheostomy hub so as to avoid torque) with adapters and pressure testing supplies. Monitor transtracheal reading/pressure testing, which measures end-expiratory pressure.
- D. Application of PMV for patients with tracheostomy tube only (without a ventilator)
- a. Physician order.
- b. SLP conducts bedside evaluation for use of PMV, in conjunction with RT, for initial placement.
- c. Pressure testing supplies (see Image 2) d . Position patient upright.
- e. Observe baseline vitals.
- f. Oral care and suctioning, as needed.
- g. RT to deep suction trach, if needed.
- h. Slowly deflate cuff.
- i. Suction trach and mouth again, as needed.
- j. Observe changes in vitals, color, work of breathing, and signs of stress.
- k. Support tracheostomy tube neck flange with one hand and gently apply PMV and transtracheal pressure manometry to the tracheostomy hub, using a gentle quarter turn twist to the right to seat the Valve on the tracheostomy hub. To remove, support the tracheostomy tube neck flange and turn to the right, while using a gentle pulling off motion.
- l. Monitor transtracheal reading/pressure testing, which measures end-expiratory pressure.
- m. Monitor stability.
- n. Pressure reading values:
- E. Signs that the patient has not tolerated the Valve
- a. Significant change in vitals with cuff deflation or Valve placement.
- b. Stress signs, such as changes in color or increased work of breathing.
- c. High-pressure testing with TTP.
- d. If a “whoosh” sound occurs when the Valve is removed following resting breaths, there is a concern for breath stacking.
- F. Additional information:
- a. The patient may cough because of an increased sensation of secretions. This type of cough is not a sign of poor PMV tolerance.
- b. Oxygen may be delivered via T-piece, trach collar, PMV oxygen adapter, or ventilator.
- c. Humidity may be provided via a tracheostomy collar or T-piece. Humidification does not affect the function of Valve.
- d. Alarms may need to be adjusted or managed by RT with physician orders.
- G. Following the PMV trial
- a. Either the SLP or the RT will document the patient’s ability to wear the Valve.
- b. The SLP, RT, and ordering physician will determine the plan for ongoing Valve trials, and the physician will write any appropriate orders.
- c. Ongoing pressure testing will likely not be completed, unless concerns are noted.
This article is from the Fall 2019 Protocol Issue of Aerodigestive Health. Click here to view Infants and Children with Tracheostomy and Ventilator Dependence.
References:
References
Abraham, S. S. (2009). Perspectives on the pediatric larynx with tracheotomy. In: Fried M., Ferlito, A. (eds): The Larynx. San Diego: Plural Publishing, Chapter 32.
Barbato, A., Bottecchia, L., & Snijders, D. (2012). Tracheostomy in children: an ancient procedure still under debate. European Respiratory Journal, 40(6), 13221323.
Barraza, G. Y., Fernandez, C., Halaby, C., Ambrosio, S., Simpser, E. F., & Pirzada, M. B. (2014). The safety of tracheostomy speaking valve use during sleep in children: A pilot study. American Journal of Otolaryngology, 35(5), 636-640.
Beard, B., & Monaco, F. J. (1993). Tracheostomy discontinuation: Impact of tube selection on resistance during tube occlusion. Respiratory Care, 38(3), 267-270.
Buswell, C., Powell, J., & Powell, S. (2016). Paediatric tracheostomy speaking valves: Our experience of forty-two children with an adapted Passy Muir® Speaking Valve. Clinical Otolaryngology, 42(4), 941-944. doi:10.1111/coa.12776
Cowell, J., Schlosser, D., & Joy, P. (2000). Language outcomes following infant tracheostomy. Asia Pacific Journal of Speech, Language, and Hearing, 5(3), 179-186. Published online 2013. http://dx.doi.org/10.1179/136132800805576942
DeMauro, S. B., D’Agostino, J. A., Bann, C., Bernbaum, J., Gerdes, M., Bell, E. F., …Kirpalani, H. (2014). Developmental outcomes of very preterm infants with tracheostomies. Journal of Pediatrics, 164(6), 1303-10.
Hernandez, G., Pedrosa, A., Ortiz, R., Accuaroni, M. D., Cuena, R., Collado, C. V., Fernandez, R. (2013). The effects of increasing effective airway diameter on weaning from mechanical ventilation in tracheostomized patients: A randomized controlled trial. Intensive Care Medicine, 39(6), 1063-1070. doi:10.1007/s00134013-2870-7
Hess, D. R. (2005). Facilitating speech in the patient with a tracheostomy. Respiratory Care, 50(4): 519-525.
Hess, D. R. & Altobelli, N. P. (2014). Tracheostomy tubes. Respiratory Care, 59 (6): 956-971. Doi: 10.4187/respcare.02920
Hofmann, L., Bolton, J., & Ferry, S. (2008). Passy-Muir speaking valve use in a children’s hospital: An interdisciplinary approach. Perspectives on Voice and Voice Disorders, 18(2): 76 – 86. Doi: 10.1044/vvd18.2.76
Hull, E. M., Dumas, H. M., Crowley, R. A., & Kharasch, V. S. (2005). Tracheostomy speaking valves for children: Tolerance and clinical benefits. Pediatric Rehabilitation, 8(3), 214-219. doi: 10.1080/13638490400021503
Keens, T., Kun, S., & Davidson Ward, S. (2017). Chronic respiratory failure. In W. Morris, K. McMillan, & D. Schaffner (eds). Roger’s Handbook of Pediatric Intensive Care. Philadelphia, PA: Walters Kluwer.
Lieu, J. E. C., Muntz, H. R., Prater, D., & Stahl, M. B. (1999). Passy-Muir Valve in children with tracheotomy. International Journal of Pediatric Otorhinolaryngology, 50(3), 197-203.
Lewis, C. W., Carron, J. D., Perkins, J. A., Sie, K. C., & Feudtner, C. (2003). Tracheotomy in pediatric patients: A national perspective. Archives of Otolaryngology–Head & Neck Surgery, 129(5), 523-529.
Mehta, A. K. & Chamyal, P. C. (1999). Tracheostomy complications and their management. Medical Journal Armed Forces India, 55 (3), 197 – 200.
O’Connor, L. R., Morris, N. R., & Paratz, J. (2019). Physiological and clinical outcomes associated with use of one-way speaking valves on tracheostomised patients: A systematic review. Heart & Lung, 48(4), 356-364. doi:10.1016/j.hrtlng.2018.11.006
Shaker, C. & Mutnik, C. (2012). Baby Trachs: Use of the Passy Muir® Valve in the NICU to Optimize Swallowing and Feeding. [self-study webinar]. www.passymuir.com/ceus
Suiter, D., McCullough, G. H., & Powell, P. W. (2003). Effects of cuff deflation and one-way tracheostomy speaking valve placement on swallowing physiology.
Dysphagia, 18(4), 284-292. doi:10.1007/s00455-003-0022-x
Sutt, A. L., Caruana, L. R., Dunster, K. R., Cornwell, P. L., Anstey, C. M., & Fraser, J. F. (2016). Speaking valves in tracheostomised ICU patients weaning off mechanical ventilation–do they facilitate lung recruitment? Journal of Critical Care, 20:91. doi: 10.1186/s13054-016-1249-x.
Torres, L. & Sirbegovic, D. J. (2004). Clinical benefits of the Passy Muir Tracheostomy and Ventilator Speaking Valves in the NICU. Neonatal Intensive Care, 17(4): 20-23.
Trotter, L. B. (1995). Success predictors for Passy Muir Speaking valve use in a pediatric population: a method evaluation.[abstract]. Respiratory Care, 40(11), 1099-1242.
Utrarachkij, J., Pongsasnongkul, J., Preutthipan, A., & Chantarojanasri, T. (2005). Measurement of end-expiratory pressure as an indicator of airway patency above tracheostomy in children. Journal of the Medical Association of Thailand, 88(7), 928-33.
Watters, K. F. (2017). Tracheostomy in infants and children. Respiratory Care, 62(6), 799-825. doi:10.4187/respcare.05366











